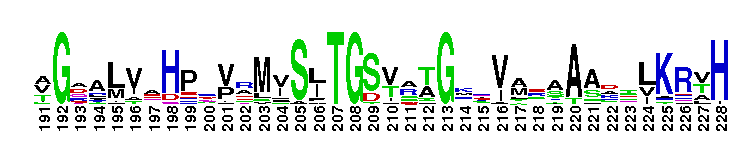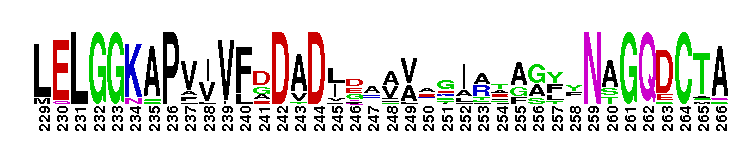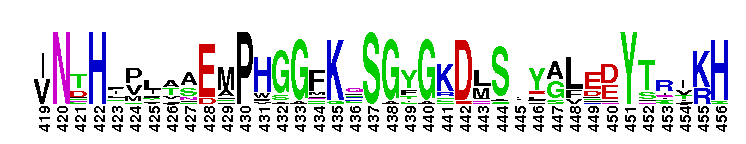| |||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




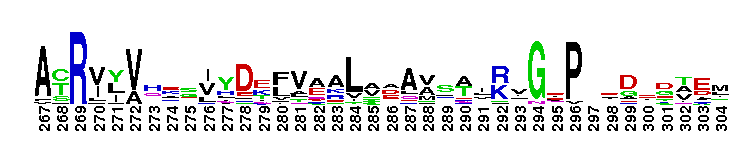
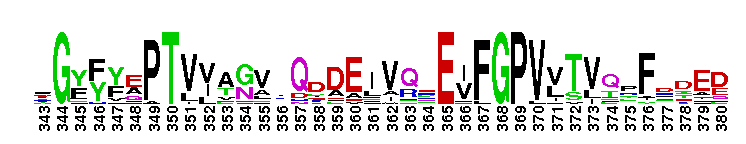
 Escherichia coli NAD+-dependent gamma-aminobutyraldehyde dehydrogenase YdcW-like. NAD+-dependent, tetrameric, gamma-aminobutyraldehyde dehydrogenase (ABALDH), YdcW of Escherichia coli K12, catalyzes the oxidation of gamma-aminobutyraldehyde to gamma-aminobutyric acid. ABALDH can also oxidize n-alkyl medium-chain aldehydes, but with a lower catalytic efficiency.
Escherichia coli NAD+-dependent gamma-aminobutyraldehyde dehydrogenase YdcW-like. NAD+-dependent, tetrameric, gamma-aminobutyraldehyde dehydrogenase (ABALDH), YdcW of Escherichia coli K12, catalyzes the oxidation of gamma-aminobutyraldehyde to gamma-aminobutyric acid. ABALDH can also oxidize n-alkyl medium-chain aldehydes, but with a lower catalytic efficiency. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.