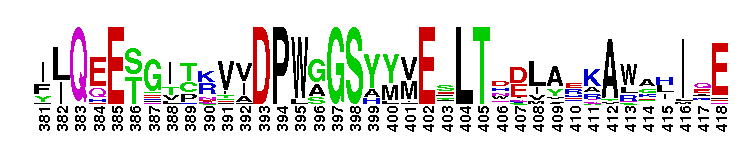
| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




 Coenzyme B12-dependent-methylmalonyl coenzyme A (CoA) mutase (MCM) family, Alpha subunit-like subfamily; contains proteins similar to the alpha subunit of Propionbacterium shermanni MCM, as well as human and E. coli MCM. Members of this subfamily contain an N-terminal MCM domain and a C-terminal coenzyme B12 binding domain. MCM catalyzes the isomerization of methylmalonyl-CoA to succinyl-CoA. The reaction proceeds via radical intermediates beginning with a substrate-induced homolytic cleavage of the Co-C bond of coenzyme B12 to produce cob(II)alamin and the deoxyadenosyl radical. MCM plays an important role in the conversion of propionyl-CoA to succinyl-CoA during the degradation of propionate for the Krebs cycle. In higher animals, MCM is involved in the breakdown of odd-chain fatty acids, several amino acids, and cholesterol. Methylobacterium extorquens MCM participates in the glyoxylate regeneration pathway. In M. extorquens, MCM forms a complex with MeaB; MeaB may protect MCM from irreversible inactivation. In some bacteria, MCM is involved in the reverse metabolic reaction, the rearrangement of succinyl-CoA to methylmalonyl-CoA. Examples include P. shermanni MCM during propionic acid fermentation, E.coli MCM in a pathway for the conversion of succinate to propionate and Streptomyces MCM in polyketide biosynthesis. Sinorhizobium meliloti strain SU47 MCM plays a role in the polyhydroxyalkanoate degradation pathway. P. shermanni and Streptomyces cinnamonensis MCMs are alpha/beta heterodimers. It has been shown for P. shermanni MCM that only the alpha subunit binds coenzyme B12 and substrates. Human MCM is a homodimer with two active sites. Mouse and E.coli MCMs are also homodimers. In humans, impaired activity of MCM results in methylmalonic aciduria, a disorder of propionic acid metabolism.
Coenzyme B12-dependent-methylmalonyl coenzyme A (CoA) mutase (MCM) family, Alpha subunit-like subfamily; contains proteins similar to the alpha subunit of Propionbacterium shermanni MCM, as well as human and E. coli MCM. Members of this subfamily contain an N-terminal MCM domain and a C-terminal coenzyme B12 binding domain. MCM catalyzes the isomerization of methylmalonyl-CoA to succinyl-CoA. The reaction proceeds via radical intermediates beginning with a substrate-induced homolytic cleavage of the Co-C bond of coenzyme B12 to produce cob(II)alamin and the deoxyadenosyl radical. MCM plays an important role in the conversion of propionyl-CoA to succinyl-CoA during the degradation of propionate for the Krebs cycle. In higher animals, MCM is involved in the breakdown of odd-chain fatty acids, several amino acids, and cholesterol. Methylobacterium extorquens MCM participates in the glyoxylate regeneration pathway. In M. extorquens, MCM forms a complex with MeaB; MeaB may protect MCM from irreversible inactivation. In some bacteria, MCM is involved in the reverse metabolic reaction, the rearrangement of succinyl-CoA to methylmalonyl-CoA. Examples include P. shermanni MCM during propionic acid fermentation, E.coli MCM in a pathway for the conversion of succinate to propionate and Streptomyces MCM in polyketide biosynthesis. Sinorhizobium meliloti strain SU47 MCM plays a role in the polyhydroxyalkanoate degradation pathway. P. shermanni and Streptomyces cinnamonensis MCMs are alpha/beta heterodimers. It has been shown for P. shermanni MCM that only the alpha subunit binds coenzyme B12 and substrates. Human MCM is a homodimer with two active sites. Mouse and E.coli MCMs are also homodimers. In humans, impaired activity of MCM results in methylmalonic aciduria, a disorder of propionic acid metabolism. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.