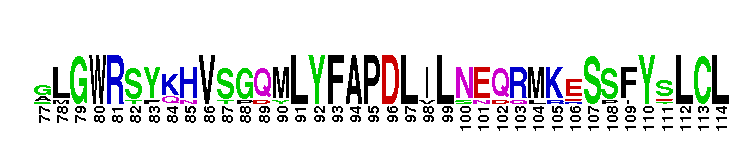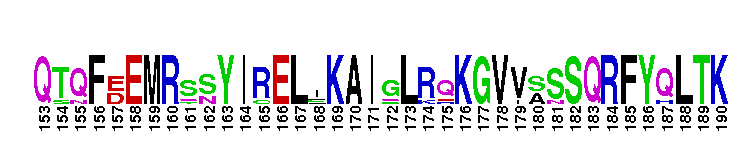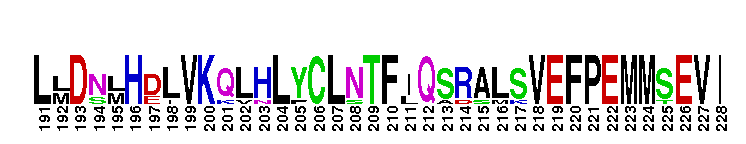| ||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




 Ligand binding domain of the progesterone receptor, a member of the nuclear hormone receptor. The ligand binding domain of the progesterone receptor (PR): PR is a member of the nuclear receptor superfamily of ligand dependent transcription factors, mediating the biological actions of progesterone. PR functions in a variety of biological processes including development of the mammary gland, regulating cell cycle progression, protein processing, and metabolism. When no binding hormone is present the carboxyl terminal inhibits transcription. Binding to a hormone induces a structural change that removes the inhibitory action. After progesterone binds to the receptor, PR forms a dimer and the complex enters the nucleus where it interacts with the hormone response element (HRE) in the promoters of progesterone responsive genes and alters their transcription. In addition, rapid actions of PR that occur independent of transcription, have also been observed in several tissues like brain, liver, mammary gland and spermatozoa. There are two natural PR isoforms called PR-A and PR-B. PR-B has an additional stretc h of 164 amino acids at the N terminus. The extra domain in PR-B performs activation functions by recruiting coactivators that could not be recruited by PR-A. Like other members of the nuclear receptor (NR) superfamily of ligand-activated transcription factors, PR has a central well conserved DNA binding domain (DBD), a variable N-terminal domain, a flexible hinge and a C-terminal ligand binding domain (LBD). The LBD is not only involved in binding to progesterone, but also involved in coactivator binding and dimerization.
Ligand binding domain of the progesterone receptor, a member of the nuclear hormone receptor. The ligand binding domain of the progesterone receptor (PR): PR is a member of the nuclear receptor superfamily of ligand dependent transcription factors, mediating the biological actions of progesterone. PR functions in a variety of biological processes including development of the mammary gland, regulating cell cycle progression, protein processing, and metabolism. When no binding hormone is present the carboxyl terminal inhibits transcription. Binding to a hormone induces a structural change that removes the inhibitory action. After progesterone binds to the receptor, PR forms a dimer and the complex enters the nucleus where it interacts with the hormone response element (HRE) in the promoters of progesterone responsive genes and alters their transcription. In addition, rapid actions of PR that occur independent of transcription, have also been observed in several tissues like brain, liver, mammary gland and spermatozoa. There are two natural PR isoforms called PR-A and PR-B. PR-B has an additional stretc h of 164 amino acids at the N terminus. The extra domain in PR-B performs activation functions by recruiting coactivators that could not be recruited by PR-A. Like other members of the nuclear receptor (NR) superfamily of ligand-activated transcription factors, PR has a central well conserved DNA binding domain (DBD), a variable N-terminal domain, a flexible hinge and a C-terminal ligand binding domain (LBD). The LBD is not only involved in binding to progesterone, but also involved in coactivator binding and dimerization. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.