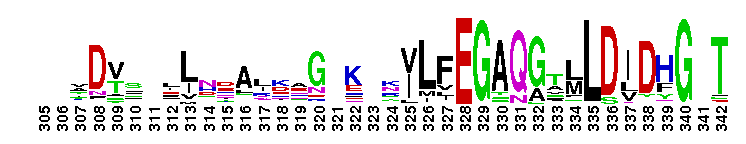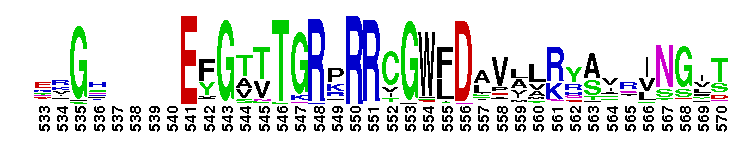| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position:  No Conserved Features/Sites Found for Adenylsucc_synt No Conserved Features/Sites Found for Adenylsucc_synt
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




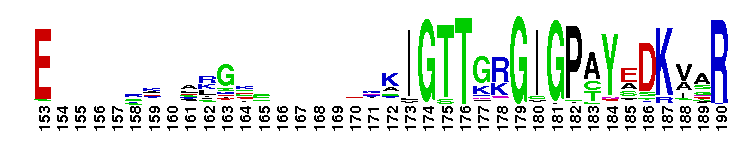
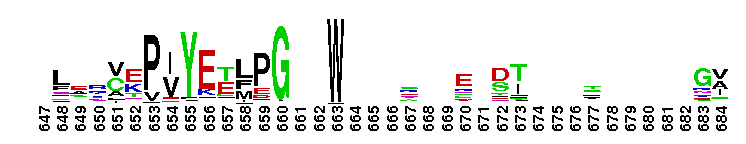
 Adenylosuccinate synthetase. Adenylosuccinate synthetase plays an important role in purine biosynthesis, by catalyzing the GTP-dependent conversion of IMP and aspartic acid to AMP. Adenylosuccinate synthetase has been characterized from various sources ranging from Escherichia coli (gene purA) to vertebrate tissues. In vertebrates, two isozymes are present - one involved in purine biosynthesis and the other in the purine nucleotide cycle. The crystal structure of adenylosuccinate synthetase from E. coli reveals that the dominant structural element of each monomer of the homodimer is a central beta-sheet of 10 strands. The first nine strands of the sheet are mutually parallel with right-handed crossover connections between the strands. The 10th strand is antiparallel with respect to the first nine strands. In addition, the enzyme has two antiparallel beta-sheets, comprised of two strands and three strands each, 11 alpha-helices and two short 3/10-helices. Further, it has been suggested that the similarities in the GTP-binding domains of the synthetase and the p21ras protein are an example of convergent evolution of two distinct families of GTP-binding proteins. Structures of adenylosuccinate synthetase from Triticum aestivum and Arabidopsis thaliana when compared with the known structures from E. coli reveals that the overall fold is very similar to that of the E. coli protein.
Adenylosuccinate synthetase. Adenylosuccinate synthetase plays an important role in purine biosynthesis, by catalyzing the GTP-dependent conversion of IMP and aspartic acid to AMP. Adenylosuccinate synthetase has been characterized from various sources ranging from Escherichia coli (gene purA) to vertebrate tissues. In vertebrates, two isozymes are present - one involved in purine biosynthesis and the other in the purine nucleotide cycle. The crystal structure of adenylosuccinate synthetase from E. coli reveals that the dominant structural element of each monomer of the homodimer is a central beta-sheet of 10 strands. The first nine strands of the sheet are mutually parallel with right-handed crossover connections between the strands. The 10th strand is antiparallel with respect to the first nine strands. In addition, the enzyme has two antiparallel beta-sheets, comprised of two strands and three strands each, 11 alpha-helices and two short 3/10-helices. Further, it has been suggested that the similarities in the GTP-binding domains of the synthetase and the p21ras protein are an example of convergent evolution of two distinct families of GTP-binding proteins. Structures of adenylosuccinate synthetase from Triticum aestivum and Arabidopsis thaliana when compared with the known structures from E. coli reveals that the overall fold is very similar to that of the E. coli protein. No pairwise interactions found for the domain Adenylsucc_synt
No pairwise interactions found for the domain Adenylsucc_synt