| |||||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




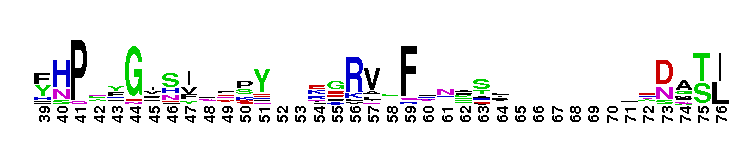
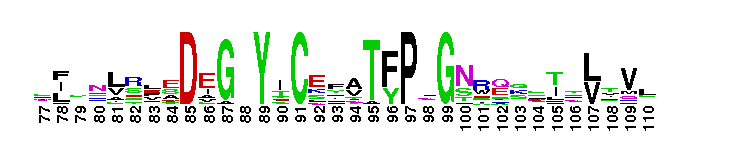
 First immunoglobulin (Ig) domain of poliovirus receptor (PVR, also known as CD155) and similar proteins. Ig1_PVR_like: domain similar to the first immunoglobulin (Ig) domain of poliovirus receptor (PVR, also known as CD155). Poliovirus (PV) binds to its cellular receptor (PVR/CD155) to initiate infection. CD155 is a membrane-anchored, single-span glycoprotein; its extracellular region has three Ig-like domains. There are four different isotypes of CD155 (referred to as alpha, beta, gamma, and delta), that result from alternate splicing of the CD155 mRNA, and have identical extracellular domains. CD155-beta and - gamma, are secreted, CD155-alpha and delta are membrane-bound and function as PV receptors. The virus recognition site is contained in the amino-terminal domain, D1. Having the virus attachment site on the receptor distal from the plasma membrane, may be important for successful initiation of infection of cells by the virus. CD155 binds in the poliovirus "canyon" with a footprint similar to that of the intercellular adhesion molecule-1 receptor on human rhinoviruses. This group also includes the first Ig-like domain of nectin-1 (also known as poliovirus receptor related protein(PVRL)1; CD111), nectin-3 (also known as PVRL 3), nectin-4 (also known as PVRL4; LNIR receptor)and DNAX accessory molecule 1 (DNAM-1; CD226).
First immunoglobulin (Ig) domain of poliovirus receptor (PVR, also known as CD155) and similar proteins. Ig1_PVR_like: domain similar to the first immunoglobulin (Ig) domain of poliovirus receptor (PVR, also known as CD155). Poliovirus (PV) binds to its cellular receptor (PVR/CD155) to initiate infection. CD155 is a membrane-anchored, single-span glycoprotein; its extracellular region has three Ig-like domains. There are four different isotypes of CD155 (referred to as alpha, beta, gamma, and delta), that result from alternate splicing of the CD155 mRNA, and have identical extracellular domains. CD155-beta and - gamma, are secreted, CD155-alpha and delta are membrane-bound and function as PV receptors. The virus recognition site is contained in the amino-terminal domain, D1. Having the virus attachment site on the receptor distal from the plasma membrane, may be important for successful initiation of infection of cells by the virus. CD155 binds in the poliovirus "canyon" with a footprint similar to that of the intercellular adhesion molecule-1 receptor on human rhinoviruses. This group also includes the first Ig-like domain of nectin-1 (also known as poliovirus receptor related protein(PVRL)1; CD111), nectin-3 (also known as PVRL 3), nectin-4 (also known as PVRL4; LNIR receptor)and DNAX accessory molecule 1 (DNAM-1; CD226). No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.