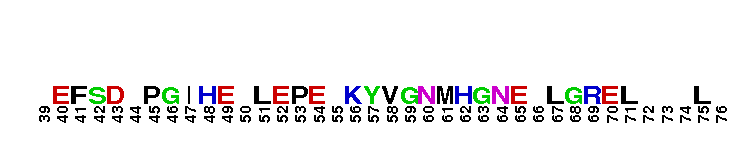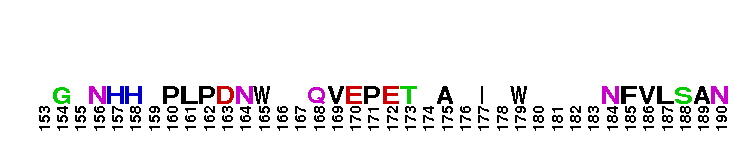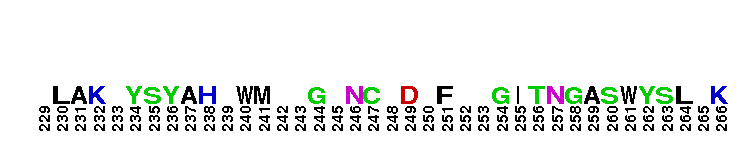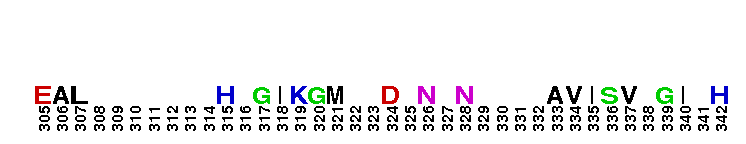| ||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




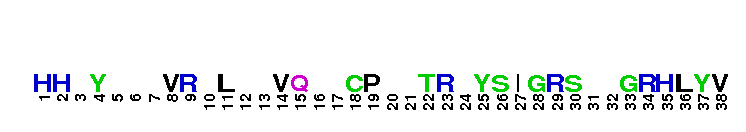
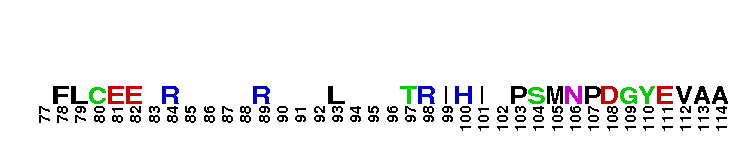
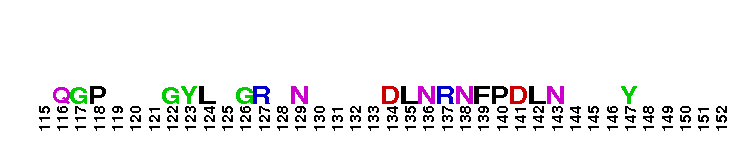
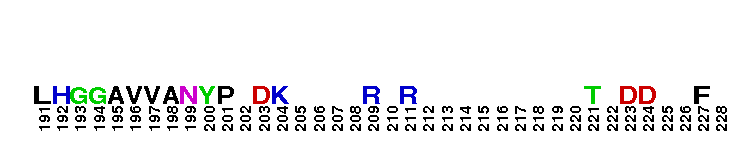
 Peptidase M14 carboxypeptidase subfamily N/E-like; Carboxypeptidase N subgroup. Peptidase M14 Carboxypeptidase N (CPN, also known as kininase I, creatine kinase conversion factor, plasma carboxypeptidase B, arginine carboxypeptidase, and protaminase; EC 3.4.17.3) is an extracellular glycoprotein synthesized in the liver and released into the blood, where it is present in high concentrations. CPN belongs to the N/E subfamily of the M14 family of metallocarboxypeptidases (MCPs).The M14 family are zinc-binding carboxypeptidases (CPs) which hydrolyze single, C-terminal amino acids from polypeptide chains, and have a recognition site for the free C-terminal carboxyl group, which is a key determinant of specificity. CPN plays an important role in protecting the body from excessive buildup of potentially deleterious peptides that normally act as local autocrine or paracrine hormones. It specifically removes C-terminal basic residues. As CPN can cleave lysine more avidly than arginine residues it is also called lysine carboxypeptidase. CPN substrates include peptides found in the bloodstream, such as kinins (e.g. bradykinin, kalinin, met-lys-bradykinin), complement anaphylatoxins and creatine kinase MM (CK-MM). By removing just one amino acid, CPN can alter peptide activity and receptor binding. For example Bradykinin, a nine-residue peptide released from kiningen in response to tissue injury which is inactivated by CPN, anaphylatoxins which are regulated by CPN by the cleaving and removal of their C-terminal arginines resulting in a reduction in their biological activities of 10-100-fold, and creatine kinase MM, a cytosolic enzyme that catalyzes the reversible transfer of a phosphate group from ATP to creatine, and is regulated by CPN by the cleavage of C-terminal lysines. Like the other N/E subfamily members, two surface loops surrounding the active-site groove restrict access to the catalytic center, thus restricting larger protein carboxypeptidase inhibitors from inhibiting CPN.
Peptidase M14 carboxypeptidase subfamily N/E-like; Carboxypeptidase N subgroup. Peptidase M14 Carboxypeptidase N (CPN, also known as kininase I, creatine kinase conversion factor, plasma carboxypeptidase B, arginine carboxypeptidase, and protaminase; EC 3.4.17.3) is an extracellular glycoprotein synthesized in the liver and released into the blood, where it is present in high concentrations. CPN belongs to the N/E subfamily of the M14 family of metallocarboxypeptidases (MCPs).The M14 family are zinc-binding carboxypeptidases (CPs) which hydrolyze single, C-terminal amino acids from polypeptide chains, and have a recognition site for the free C-terminal carboxyl group, which is a key determinant of specificity. CPN plays an important role in protecting the body from excessive buildup of potentially deleterious peptides that normally act as local autocrine or paracrine hormones. It specifically removes C-terminal basic residues. As CPN can cleave lysine more avidly than arginine residues it is also called lysine carboxypeptidase. CPN substrates include peptides found in the bloodstream, such as kinins (e.g. bradykinin, kalinin, met-lys-bradykinin), complement anaphylatoxins and creatine kinase MM (CK-MM). By removing just one amino acid, CPN can alter peptide activity and receptor binding. For example Bradykinin, a nine-residue peptide released from kiningen in response to tissue injury which is inactivated by CPN, anaphylatoxins which are regulated by CPN by the cleaving and removal of their C-terminal arginines resulting in a reduction in their biological activities of 10-100-fold, and creatine kinase MM, a cytosolic enzyme that catalyzes the reversible transfer of a phosphate group from ATP to creatine, and is regulated by CPN by the cleavage of C-terminal lysines. Like the other N/E subfamily members, two surface loops surrounding the active-site groove restrict access to the catalytic center, thus restricting larger protein carboxypeptidase inhibitors from inhibiting CPN. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.