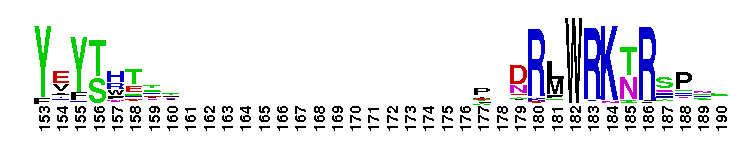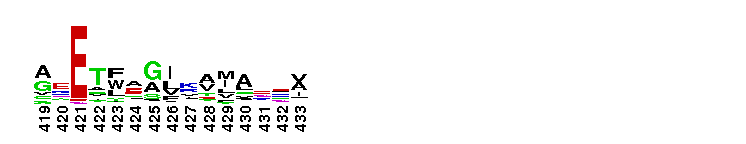| ||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




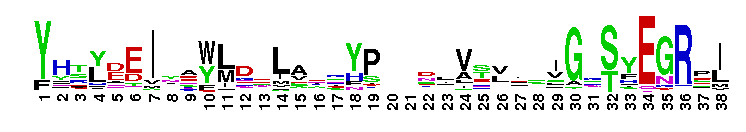
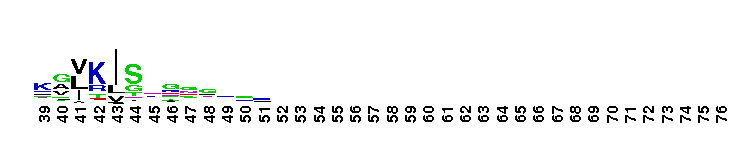
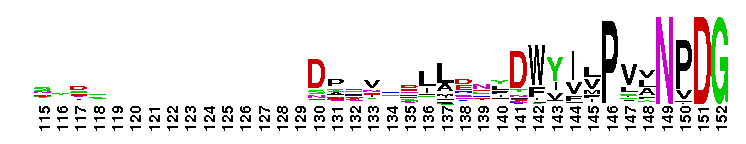
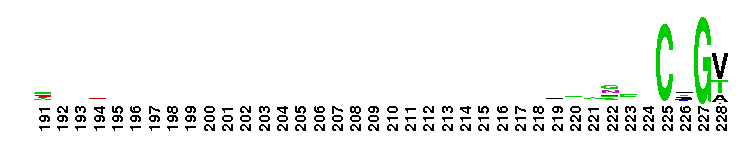
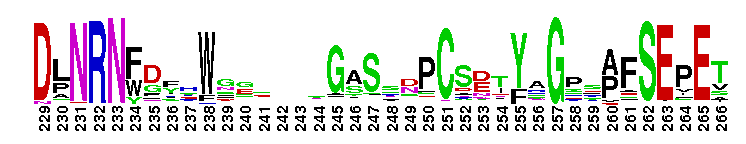
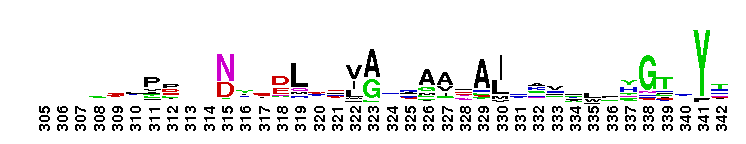
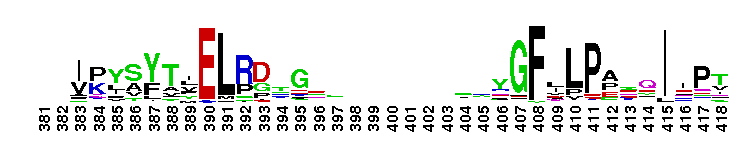
 Peptidase M14 carboxypeptidase subfamily A/B-like. The Peptidase M14 Carboxypeptidase (CP) A/B subfamily is one of two main M14 CP subfamilies defined by sequence and structural homology, the other being the N/E subfamily. CPs hydrolyze single, C-terminal amino acids from polypeptide chains. They have a recognition site for the free C-terminal carboxyl group, which is a key determinant of specificity. Enzymes belonging to the A/B subfamily are normally synthesized as inactive precursors containing preceding signal peptide, followed by a globular N-terminal pro-region linked to the enzyme; these proenzymes are called procarboxypeptidases. The A/B enzymes can be further divided based on their substrate specificity; Carboxypeptidase A-like (CPA-like) enzymes favor hydrophobic residues while carboxypeptidase B-like (CPB-like) enzymes only cleave the basic residues lysine or arginine. There are nine members in the A/B family: CPA1, CPA2, CPA3, CPA4, CPA5, CPA6, CPB, CPO and CPU. CPA1, CPA2 and CPB are produced by the pancreas. The A forms have slightly different specificities, with CPA1 preferring aliphatic and small aromatic residues, and CPA2 preferring the bulkier aromatic side chains. CPA3 is found in secretory granules of mast cells and functions in inflammatory processes. CPA4 is detected in hormone-regulated tissues, and is thought to play a role in prostate cancer. CPA5 is present in discrete regions of pituitary and other tissues, and cleaves aliphatic C-terminal residues. CPA6 is highly expressed in embryonic brain and optic muscle, suggesting that it may play a specific role in cell migration and axonal guidance. CPU (also called CPB2) is produced and secreted by the liver as the inactive precursor, PCPU, commonly referred to as thrombin-activatable fibrinolysis inhibitor (TAFI). Little is known about CPO but it has been suggested to have specificity for acidic residues.
Peptidase M14 carboxypeptidase subfamily A/B-like. The Peptidase M14 Carboxypeptidase (CP) A/B subfamily is one of two main M14 CP subfamilies defined by sequence and structural homology, the other being the N/E subfamily. CPs hydrolyze single, C-terminal amino acids from polypeptide chains. They have a recognition site for the free C-terminal carboxyl group, which is a key determinant of specificity. Enzymes belonging to the A/B subfamily are normally synthesized as inactive precursors containing preceding signal peptide, followed by a globular N-terminal pro-region linked to the enzyme; these proenzymes are called procarboxypeptidases. The A/B enzymes can be further divided based on their substrate specificity; Carboxypeptidase A-like (CPA-like) enzymes favor hydrophobic residues while carboxypeptidase B-like (CPB-like) enzymes only cleave the basic residues lysine or arginine. There are nine members in the A/B family: CPA1, CPA2, CPA3, CPA4, CPA5, CPA6, CPB, CPO and CPU. CPA1, CPA2 and CPB are produced by the pancreas. The A forms have slightly different specificities, with CPA1 preferring aliphatic and small aromatic residues, and CPA2 preferring the bulkier aromatic side chains. CPA3 is found in secretory granules of mast cells and functions in inflammatory processes. CPA4 is detected in hormone-regulated tissues, and is thought to play a role in prostate cancer. CPA5 is present in discrete regions of pituitary and other tissues, and cleaves aliphatic C-terminal residues. CPA6 is highly expressed in embryonic brain and optic muscle, suggesting that it may play a specific role in cell migration and axonal guidance. CPU (also called CPB2) is produced and secreted by the liver as the inactive precursor, PCPU, commonly referred to as thrombin-activatable fibrinolysis inhibitor (TAFI). Little is known about CPO but it has been suggested to have specificity for acidic residues. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.