| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




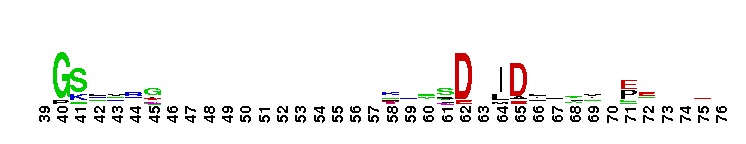
 Nucleotidyltransferase (NT) domain of DNA polymerase beta and similar proteins. This superfamily includes the NT domains of DNA polymerase beta and other family X DNA polymerases, as well as the NT domains of Class I and Class II CCA-adding enzymes, RelA- and SpoT-like ppGpp synthetases and hydrolases, 2'5'-oligoadenylate (2-5A)synthetases, Escherichia coli adenylyltransferase (GlnE), Escherichia coli uridylyl transferase (GlnD), poly (A) polymerases, terminal uridylyl transferases, and Staphylococcus aureus kanamycin nucleotidyltransferase, and similar proteins. The Escherichia coli CCA-adding enzyme belongs to this superfamily but is not included as this enzyme lacks the N-terminal helix conserved in the remainder of the superfamily. In the majority of the Pol beta-like superfamily NTs, two carboxylates, Dx[D/E], together with a third more distal carboxylate coordinate two divalent metal cations that are essential for catalysis. These divalent metal ions are involved in a two-metal ion mechanism of nucleotide addition. Two of the three catalytic carboxylates are found in Rel-Spo enzymes, with the second carboxylate of the DXD motif missing. Evidence supports a single-cation synthetase mechanism for Rel-Spo enzymes.
Nucleotidyltransferase (NT) domain of DNA polymerase beta and similar proteins. This superfamily includes the NT domains of DNA polymerase beta and other family X DNA polymerases, as well as the NT domains of Class I and Class II CCA-adding enzymes, RelA- and SpoT-like ppGpp synthetases and hydrolases, 2'5'-oligoadenylate (2-5A)synthetases, Escherichia coli adenylyltransferase (GlnE), Escherichia coli uridylyl transferase (GlnD), poly (A) polymerases, terminal uridylyl transferases, and Staphylococcus aureus kanamycin nucleotidyltransferase, and similar proteins. The Escherichia coli CCA-adding enzyme belongs to this superfamily but is not included as this enzyme lacks the N-terminal helix conserved in the remainder of the superfamily. In the majority of the Pol beta-like superfamily NTs, two carboxylates, Dx[D/E], together with a third more distal carboxylate coordinate two divalent metal cations that are essential for catalysis. These divalent metal ions are involved in a two-metal ion mechanism of nucleotide addition. Two of the three catalytic carboxylates are found in Rel-Spo enzymes, with the second carboxylate of the DXD motif missing. Evidence supports a single-cation synthetase mechanism for Rel-Spo enzymes. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.