| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




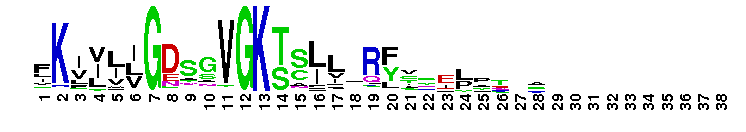
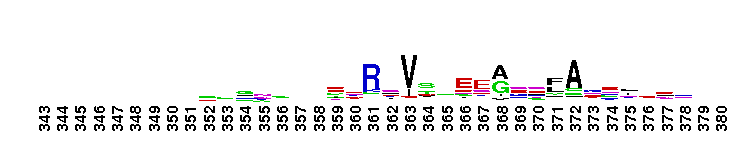
 Ras-related in brain (Rab) family of small guanosine triphosphatases (GTPases). Rab GTPases form the largest family within the Ras superfamily. There are at least 60 Rab genes in the human genome, and a number of Rab GTPases are conserved from yeast to humans. Rab GTPases are small, monomeric proteins that function as molecular switches to regulate vesicle trafficking pathways. The different Rab GTPases are localized to the cytosolic face of specific intracellular membranes, where they regulate distinct steps in membrane traffic pathways. In the GTP-bound form, Rab GTPases recruit specific sets of effector proteins onto membranes. Through their effectors, Rab GTPases regulate vesicle formation, actin- and tubulin-dependent vesicle movement, and membrane fusion. GTPase activating proteins (GAPs) interact with GTP-bound Rab and accelerate the hydrolysis of GTP to GDP. Guanine nucleotide exchange factors (GEFs) interact with GDP-bound Rabs to promote the formation of the GTP-bound state. Rabs are further regulated by guanine nucleotide dissociation inhibitors (GDIs), which mask C-terminal lipid binding and promote cytosolic localization. While most unicellular organisms possess 5-20 Rab members, several have been found to possess 60 or more Rabs; for many of these Rab isoforms, homologous proteins are not found in other organisms. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins. Since crystal structures often lack C-terminal residues, the lipid modification site is not available for annotation in many of the CDs in the hierarchy, but is included where possible.
Ras-related in brain (Rab) family of small guanosine triphosphatases (GTPases). Rab GTPases form the largest family within the Ras superfamily. There are at least 60 Rab genes in the human genome, and a number of Rab GTPases are conserved from yeast to humans. Rab GTPases are small, monomeric proteins that function as molecular switches to regulate vesicle trafficking pathways. The different Rab GTPases are localized to the cytosolic face of specific intracellular membranes, where they regulate distinct steps in membrane traffic pathways. In the GTP-bound form, Rab GTPases recruit specific sets of effector proteins onto membranes. Through their effectors, Rab GTPases regulate vesicle formation, actin- and tubulin-dependent vesicle movement, and membrane fusion. GTPase activating proteins (GAPs) interact with GTP-bound Rab and accelerate the hydrolysis of GTP to GDP. Guanine nucleotide exchange factors (GEFs) interact with GDP-bound Rabs to promote the formation of the GTP-bound state. Rabs are further regulated by guanine nucleotide dissociation inhibitors (GDIs), which mask C-terminal lipid binding and promote cytosolic localization. While most unicellular organisms possess 5-20 Rab members, several have been found to possess 60 or more Rabs; for many of these Rab isoforms, homologous proteins are not found in other organisms. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins. Since crystal structures often lack C-terminal residues, the lipid modification site is not available for annotation in many of the CDs in the hierarchy, but is included where possible. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.