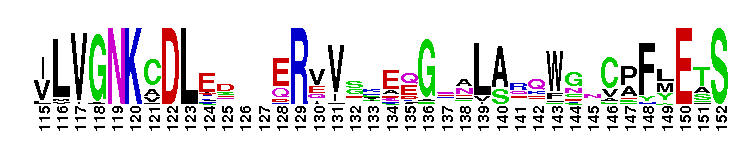| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




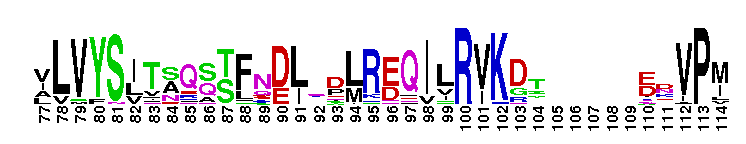
 Rap-like family consists of Rap1, Rap2 and RSR1. The Rap subfamily consists of the Rap1, Rap2, and RSR1. Rap subfamily proteins perform different cellular functions, depending on the isoform and its subcellular localization. For example, in rat salivary gland, neutrophils, and platelets, Rap1 localizes to secretory granules and is believed to regulate exocytosis or the formation of secretory granules. Rap1 has also been shown to localize in the Golgi of rat fibroblasts, zymogen granules, plasma membrane, and microsomal membrane of the pancreatic acini, as well as in the endocytic compartment of skeletal muscle cells and fibroblasts. Rap1 localizes in the nucleus of human oropharyngeal squamous cell carcinomas (SCCs) and cell lines. Rap1 plays a role in phagocytosis by controlling the binding of adhesion receptors (typically integrins) to their ligands. In yeast, Rap1 has been implicated in multiple functions, including activation and silencing of transcription and maintenance of telomeres. Rap2 is involved in multiple functions, including activation of c-Jun N-terminal kinase (JNK) to regulate the actin cytoskeleton and activation of the Wnt/beta-catenin signaling pathway in embryonic Xenopus. A number of effector proteins for Rap2 have been identified, including isoform 3 of the human mitogen-activated protein kinase kinase kinase kinase 4 (MAP4K4) and Traf2- and Nck-interacting kinase (TNIK), and the RalGEFs RalGDS, RGL, and Rlf, which also interact with Rap1 and Ras. RSR1 is the fungal homolog of Rap1 and Rap2. In budding yeasts, it is involved in selecting a site for bud growth, which directs the establishment of cell polarization. The Rho family GTPase Cdc42 and its GEF, Cdc24, then establish an axis of polarized growth. It is believed that Cdc42 interacts directly with RSR1 in vivo. In filamentous fungi such as Ashbya gossypii, RSR1 is a key regulator of polar growth in the hypha. Most Ras proteins contain a lipid modification site at the C-terminus, with a typical sequence motif CaaX, where a = an aliphatic amino acid and X = any amino acid. Lipid binding is essential for membrane attachment, a key feature of most Ras proteins. Due to the presence of truncated sequences in this CD, the lipid modification site is not available for annotation.
Rap-like family consists of Rap1, Rap2 and RSR1. The Rap subfamily consists of the Rap1, Rap2, and RSR1. Rap subfamily proteins perform different cellular functions, depending on the isoform and its subcellular localization. For example, in rat salivary gland, neutrophils, and platelets, Rap1 localizes to secretory granules and is believed to regulate exocytosis or the formation of secretory granules. Rap1 has also been shown to localize in the Golgi of rat fibroblasts, zymogen granules, plasma membrane, and microsomal membrane of the pancreatic acini, as well as in the endocytic compartment of skeletal muscle cells and fibroblasts. Rap1 localizes in the nucleus of human oropharyngeal squamous cell carcinomas (SCCs) and cell lines. Rap1 plays a role in phagocytosis by controlling the binding of adhesion receptors (typically integrins) to their ligands. In yeast, Rap1 has been implicated in multiple functions, including activation and silencing of transcription and maintenance of telomeres. Rap2 is involved in multiple functions, including activation of c-Jun N-terminal kinase (JNK) to regulate the actin cytoskeleton and activation of the Wnt/beta-catenin signaling pathway in embryonic Xenopus. A number of effector proteins for Rap2 have been identified, including isoform 3 of the human mitogen-activated protein kinase kinase kinase kinase 4 (MAP4K4) and Traf2- and Nck-interacting kinase (TNIK), and the RalGEFs RalGDS, RGL, and Rlf, which also interact with Rap1 and Ras. RSR1 is the fungal homolog of Rap1 and Rap2. In budding yeasts, it is involved in selecting a site for bud growth, which directs the establishment of cell polarization. The Rho family GTPase Cdc42 and its GEF, Cdc24, then establish an axis of polarized growth. It is believed that Cdc42 interacts directly with RSR1 in vivo. In filamentous fungi such as Ashbya gossypii, RSR1 is a key regulator of polar growth in the hypha. Most Ras proteins contain a lipid modification site at the C-terminus, with a typical sequence motif CaaX, where a = an aliphatic amino acid and X = any amino acid. Lipid binding is essential for membrane attachment, a key feature of most Ras proteins. Due to the presence of truncated sequences in this CD, the lipid modification site is not available for annotation. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.