| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




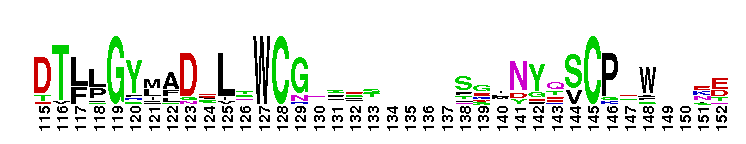
 ADP-ribosyl cyclase, also known as cyclic ADP-ribose hydrolase or CD38. ADP-ribosyl cyclase (EC:3.2.2.5) synthesizes the second messenger cyclic-ADP ribose (cADPR), which in turn releases calcium from internal stores. Mammals possess two membrane proteins, CD38 and BST-1/CD157, which exhibit ADP-ribosyl cyclase activity, as well as intracellular soluble ADP-ribose cyclases. CD38 is involved in differentiation, adhesion, and cell proliferation, and has been implicated in diseases such as AIDS, diabetes, and B-cell chronic lymphocytic leukemia. The extramembrane domain of CD38 acts as a multifunctional enzyme, and can synthesize cADPR from NAD+, hydrolyze NAD+ and cADPR to ADPR, as well as catalyze the exchange of the nicotinamide group of NADP+ with nicotinic acid under acidic conditions, to yield NAADP+ (nicotinic acid-adenine dinucleotide phosphate), a metabolite involved in Ca2+ mobilization from acidic stores.
ADP-ribosyl cyclase, also known as cyclic ADP-ribose hydrolase or CD38. ADP-ribosyl cyclase (EC:3.2.2.5) synthesizes the second messenger cyclic-ADP ribose (cADPR), which in turn releases calcium from internal stores. Mammals possess two membrane proteins, CD38 and BST-1/CD157, which exhibit ADP-ribosyl cyclase activity, as well as intracellular soluble ADP-ribose cyclases. CD38 is involved in differentiation, adhesion, and cell proliferation, and has been implicated in diseases such as AIDS, diabetes, and B-cell chronic lymphocytic leukemia. The extramembrane domain of CD38 acts as a multifunctional enzyme, and can synthesize cADPR from NAD+, hydrolyze NAD+ and cADPR to ADPR, as well as catalyze the exchange of the nicotinamide group of NADP+ with nicotinic acid under acidic conditions, to yield NAADP+ (nicotinic acid-adenine dinucleotide phosphate), a metabolite involved in Ca2+ mobilization from acidic stores. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.