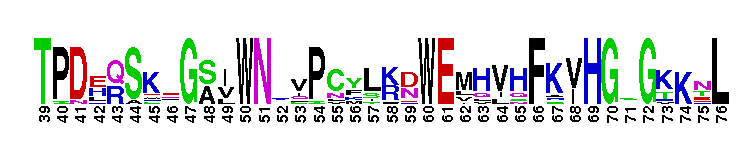| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




 VIP36 and VIPL type 1 transmembrane proteins, lectin domain. The vesicular integral protein of 36 kDa (VIP36) is a type 1 transmembrane protein of the mammalian early secretory pathway that acts as a cargo receptor transporting high mannose type glycoproteins between the Golgi and the endoplasmic reticulum (ER). Lectins of the early secretory pathway are involved in the selective transport of newly synthesized glycoproteins from the ER to the ER-Golgi intermediate compartment (ERGIC). The most prominent cycling lectin is the mannose-binding type1 membrane protein ERGIC-53, which functions as a cargo receptor to facilitate export of glycoproteins from the ER. L-type lectins have a dome-shaped beta-barrel carbohydrate recognition domain with a curved seven-stranded beta-sheet referred to as the "front face" and a flat six-stranded beta-sheet referred to as the "back face". This domain homodimerizes so that adjacent back sheets form a contiguous 12-stranded sheet and homotetramers occur by a back-to-back association of these homodimers. Though L-type lectins exhibit both sequence and structural similarity to one another, their carbohydrate binding specificities differ widely.
VIP36 and VIPL type 1 transmembrane proteins, lectin domain. The vesicular integral protein of 36 kDa (VIP36) is a type 1 transmembrane protein of the mammalian early secretory pathway that acts as a cargo receptor transporting high mannose type glycoproteins between the Golgi and the endoplasmic reticulum (ER). Lectins of the early secretory pathway are involved in the selective transport of newly synthesized glycoproteins from the ER to the ER-Golgi intermediate compartment (ERGIC). The most prominent cycling lectin is the mannose-binding type1 membrane protein ERGIC-53, which functions as a cargo receptor to facilitate export of glycoproteins from the ER. L-type lectins have a dome-shaped beta-barrel carbohydrate recognition domain with a curved seven-stranded beta-sheet referred to as the "front face" and a flat six-stranded beta-sheet referred to as the "back face". This domain homodimerizes so that adjacent back sheets form a contiguous 12-stranded sheet and homotetramers occur by a back-to-back association of these homodimers. Though L-type lectins exhibit both sequence and structural similarity to one another, their carbohydrate binding specificities differ widely. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.