| |||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




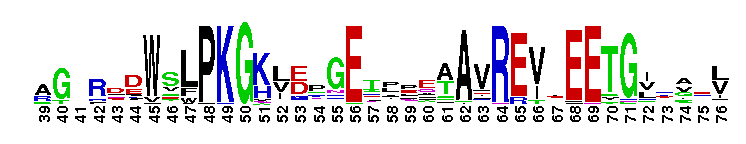
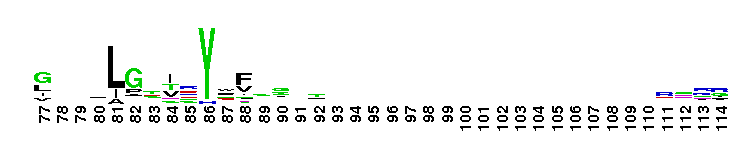
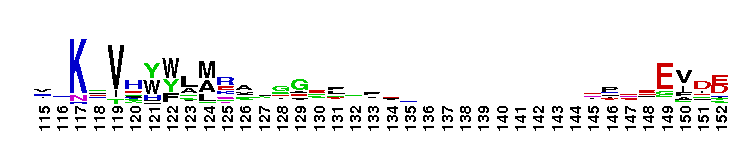
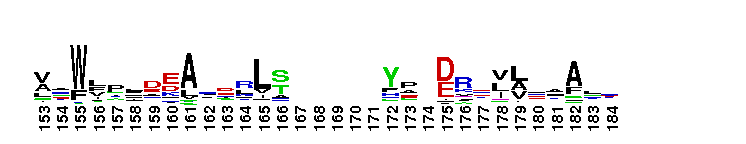
 Diadenosine hexaphosphate (Ap6A) hydrolase is a member of the Nudix hydrolase superfamily. Ap6A hydrolase specifically hydrolyzes diadenosine polyphosphates, but not ATP or diadenosine triphosphate, and it generates ATP as the product. Ap6A, the most preferred substrate, hydrolyzes to produce two ATP molecules, which is a novel hydrolysis mode for Ap6A. These results indicate that Ap6A hydrolase is a diadenosine polyphosphate hydrolase. It requires the presence of a divalent cation, such as Mn2+, Mg2+, Zn2+, and Co2+, for activity. Members of the Nudix superfamily are recognized by a highly conserved 23-residue nudix motif (GX5EX7REUXEEXGU, where U = I, L or V), which forms a structural motif that functions as a metal binding and catalytic site.
Diadenosine hexaphosphate (Ap6A) hydrolase is a member of the Nudix hydrolase superfamily. Ap6A hydrolase specifically hydrolyzes diadenosine polyphosphates, but not ATP or diadenosine triphosphate, and it generates ATP as the product. Ap6A, the most preferred substrate, hydrolyzes to produce two ATP molecules, which is a novel hydrolysis mode for Ap6A. These results indicate that Ap6A hydrolase is a diadenosine polyphosphate hydrolase. It requires the presence of a divalent cation, such as Mn2+, Mg2+, Zn2+, and Co2+, for activity. Members of the Nudix superfamily are recognized by a highly conserved 23-residue nudix motif (GX5EX7REUXEEXGU, where U = I, L or V), which forms a structural motif that functions as a metal binding and catalytic site. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.