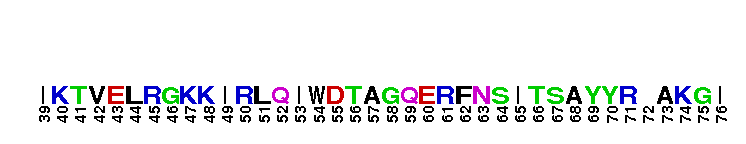| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




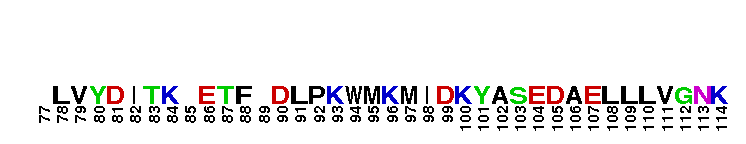
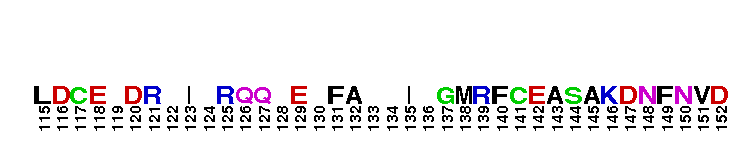
 Rab GTPase family 12 (Rab12). Rab12 was first identified in canine cells, where it was localized to the Golgi complex. The specific function of Rab12 remains unknown, and inconsistent results about its cellular localization have been reported. More recent studies have identified Rab12 associated with post-Golgi vesicles, or with other small vesicle-like structures but not with the Golgi complex. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins. GTPase activating proteins (GAPs) interact with GTP-bound Rab and accelerate the hydrolysis of GTP to GDP. Guanine nucleotide exchange factors (GEFs) interact with GDP-bound Rabs to promote the formation of the GTP-bound state. Rabs are further regulated by guanine nucleotide dissociation inhibitors (GDIs), which facilitate Rab recycling by masking C-terminal lipid binding and promoting cytosolic localization. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins.
Rab GTPase family 12 (Rab12). Rab12 was first identified in canine cells, where it was localized to the Golgi complex. The specific function of Rab12 remains unknown, and inconsistent results about its cellular localization have been reported. More recent studies have identified Rab12 associated with post-Golgi vesicles, or with other small vesicle-like structures but not with the Golgi complex. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins. GTPase activating proteins (GAPs) interact with GTP-bound Rab and accelerate the hydrolysis of GTP to GDP. Guanine nucleotide exchange factors (GEFs) interact with GDP-bound Rabs to promote the formation of the GTP-bound state. Rabs are further regulated by guanine nucleotide dissociation inhibitors (GDIs), which facilitate Rab recycling by masking C-terminal lipid binding and promoting cytosolic localization. Most Rab GTPases contain a lipid modification site at the C-terminus, with sequence motifs CC, CXC, or CCX. Lipid binding is essential for membrane attachment, a key feature of most Rab proteins. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.