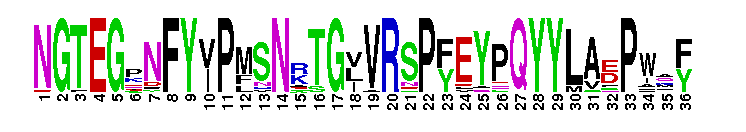| |||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position:  No Conserved Features/Sites Found for Rhodopsin_N No Conserved Features/Sites Found for Rhodopsin_N
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




 Amino terminal of the G-protein receptor rhodopsin. Rhodopsin is the archetypal G-protein-coupled receptor. Such receptors participate in virtually all physiological processes, as signalling molecules. They utilise heterotrimeric guanosine triphosphate (GTP)-binding proteins to transduce extracellular signals to intracellular events. Rhodopsin is important because of the pivotal role it plays in visual signal transduction. Rhodopsin is a dimeric transmembrane protein and its intradiskal surface consists of this amino terminal domain and three loops connecting six of the seven transmembrane helices. The N-terminus is a compact domain of alpha-helical regions with breaks and bends at proline residues outside the membrane. The transmembrane part of rhodopsin is represented by 7tm_1 (pfam00001). The N-terminal domain is extracellular is and is necessary for successful dimerisation and molecular stability.
Amino terminal of the G-protein receptor rhodopsin. Rhodopsin is the archetypal G-protein-coupled receptor. Such receptors participate in virtually all physiological processes, as signalling molecules. They utilise heterotrimeric guanosine triphosphate (GTP)-binding proteins to transduce extracellular signals to intracellular events. Rhodopsin is important because of the pivotal role it plays in visual signal transduction. Rhodopsin is a dimeric transmembrane protein and its intradiskal surface consists of this amino terminal domain and three loops connecting six of the seven transmembrane helices. The N-terminus is a compact domain of alpha-helical regions with breaks and bends at proline residues outside the membrane. The transmembrane part of rhodopsin is represented by 7tm_1 (pfam00001). The N-terminal domain is extracellular is and is necessary for successful dimerisation and molecular stability. No pairwise interactions found for the domain Rhodopsin_N
No pairwise interactions found for the domain Rhodopsin_N