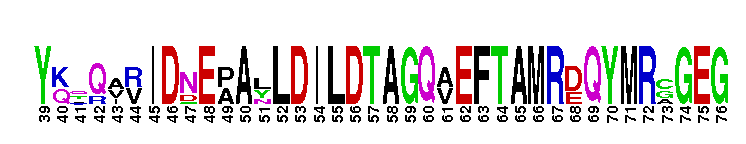| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tips:  Range on the Protein: Protein ID Protein Position Domain Position: 
|
|---|
Weblogos are Copyright (c) 2002 Regents of the University of California
| DMDM_info@umbc.edu | 1000 Hilltop Circle, Baltimore, MD 21250 | Department of Biological Sciences | Phone: 410-455-2258 |




 Ras-like protein in all tissues (Rit), Ras-like protein in neurons (Rin) and Ras-related protein which interacts with calmodulin (Ric). Rit (Ras-like protein in all tissues), Rin (Ras-like protein in neurons) and Ric (Ras-related protein which interacts with calmodulin) form a subfamily with several unique structural and functional characteristics. These proteins all lack a the C-terminal CaaX lipid-binding motif typical of Ras family proteins, and Rin and Ric contain calmodulin-binding domains. Rin, which is expressed only in neurons, induces neurite outgrowth in rat pheochromocytoma cells through its association with calmodulin and its activation of endogenous Rac/cdc42. Rit, which is ubiquitously expressed in mammals, inhibits growth-factor withdrawl-mediated apoptosis and induces neurite extension in pheochromocytoma cells. Rit and Rin are both able to form a ternary complex with PAR6, a cell polarity-regulating protein, and Rac/cdc42. This ternary complex is proposed to have physiological function in processes such as tumorigenesis. Activated Ric is likely to signal in parallel with the Ras pathway or stimulate the Ras pathway at some upstream point, and binding of calmodulin to Ric may negatively regulate Ric activity.
Ras-like protein in all tissues (Rit), Ras-like protein in neurons (Rin) and Ras-related protein which interacts with calmodulin (Ric). Rit (Ras-like protein in all tissues), Rin (Ras-like protein in neurons) and Ric (Ras-related protein which interacts with calmodulin) form a subfamily with several unique structural and functional characteristics. These proteins all lack a the C-terminal CaaX lipid-binding motif typical of Ras family proteins, and Rin and Ric contain calmodulin-binding domains. Rin, which is expressed only in neurons, induces neurite outgrowth in rat pheochromocytoma cells through its association with calmodulin and its activation of endogenous Rac/cdc42. Rit, which is ubiquitously expressed in mammals, inhibits growth-factor withdrawl-mediated apoptosis and induces neurite extension in pheochromocytoma cells. Rit and Rin are both able to form a ternary complex with PAR6, a cell polarity-regulating protein, and Rac/cdc42. This ternary complex is proposed to have physiological function in processes such as tumorigenesis. Activated Ric is likely to signal in parallel with the Ras pathway or stimulate the Ras pathway at some upstream point, and binding of calmodulin to Ric may negatively regulate Ric activity. No pairwise interactions are available for this conserved domain.
No pairwise interactions are available for this conserved domain.